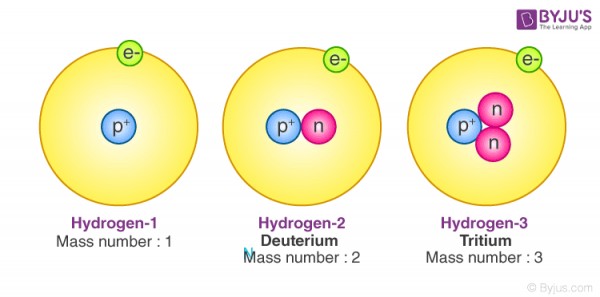
To draw an element, you will need to know what element it is. Lets use Hydrogen for example. You start with how many protons there are. This is the atomic number. So for hydrogen, the atomic number is 1. Then you will need to know how many nutrons there are. And for that, you need to know the stable forms of hydrogren. There are many isotopes for hydrogen, but I'm just going to show you the two most stable. Isotopes are forms of the element. The two isotopes are Hydrogen-1 (Protium), and Hydrogen-2 (Deuterium). Hydrogen-1 doesn't have any nutrons, so you will only need to draw the one proton and no nutrons. Hydrogen-2 has one nutron so you would need to draw one nutron if you were to draw Hydrogen-2. They are very very very small, but make sure you label them correctly. Finally, you will need to draw the electrons, which are the particles that fly around the atom. There is only one electron. If you were to go and dig a bit deeper, you would find that these electrons are on different energy levels, but for this I will keep it simple. It is the first energy level. So put one small electron in orbit around the nucleus of the atom.